02-02-2016
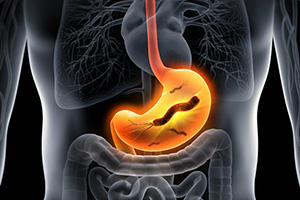
Saccharomyces boulardii: helps protect, prevent and treat various forms of diarrhoea
 Saccharomyces boulardii is a non-pathogenic, probiotic yeast. It has a decades-long history of use in Europe as an anti-diarrhoea treatment, where it is used for preventing diarrhoea associated with antibiotic treatment or nasogastric feeding, as well as for treating Clostridium difficile-induced acute diarrhoea in adults and children, and chronic diarrhoea in HIV patients.
Saccharomyces boulardii is a non-pathogenic, probiotic yeast. It has a decades-long history of use in Europe as an anti-diarrhoea treatment, where it is used for preventing diarrhoea associated with antibiotic treatment or nasogastric feeding, as well as for treating Clostridium difficile-induced acute diarrhoea in adults and children, and chronic diarrhoea in HIV patients.
Saccharomyces boulardii is a wild, tropical yeast discovered in Indochina in 1923 by French scientist Henri Boulard. Noting that the skin of various tropical fruits (lychees, mangosteens) was used by the local population to treat diarrhoea, he successfully isolated the yeast from these fruits.
In humans, Saccharomyces boulardii does not permanently colonise the intestinal tract – once supplementation ceases, cells are undetectable in stools after 6 to 7 days.
Prevention of antibiotic-associated diarrhoea
Three large-scale studies have been conducted on the preventive effects of Saccharomyces boulardii against antibiotic-associated diarrhoea. One such multi-centre trial monitored a total of 388 outpatients aged at least 15 years, across 25 centres. The subjects involved in this double-blind, placebo-controlled study had no intestinal pathology at the outset. They were given antibiotics (tetracycline or beta-lactam) for at least five days and also randomly allocated either 100mg of Saccharomyces boulardii or a placebo. Researchers found that significantly fewer of the probiotic patients went on to develop diarrhoea than those taking placebo (4.5% compared with 17.5%)1.Treating Clostridium difficile infections
Clostridium difficile is linked to a third of cases of antibiotic-related diarrhoea and almost 99.8% of pseudomembranous colitis. It is involved in a wide range of gastrointestinal diseases, from simple diarrhoea to pseudomembranous colitis and can be fatal. It is the most common cause of nosocomial (hospital-acquired) diarrhoea.Treatment of these diseases includes stopping the antibiotic medication responsible, and administering vancomycin, metronidazole or bacitracin. Unfortunately however, 10-20% of patients treated in this way suffer a relapse and are then more likely to experience repeated episodes of the disease. Clinical studies have been conducted to assess the efficacy of Saccharomyces boulardii as a treatment or adjunct treatment for Clostridium difficile.
An open-label trial examined the benefits of Saccharomyces boulardii combined with 500mg/day of vancomycin in 16 patients with recurrent disease. They were treated for 10 days with vancomycin and then for a further 30 days with 1g/day of Saccharomyces boulardii. Researchers found that 85% of subjects responded well to the combined treatment and did not suffer any relapse 2.
While young children rarely get pseudomembranous colitis, this does not mean they are invulnerable to infection from Clostridium difficile. In one study carried out on 19 babies and children with chronic diarrhoea, Clostridium difficile toxin B was the only pathogenic germ identified in culture of their faeces. The majority of the children had recently undergone antibiotic treatment. A 15-day course of treatment with Saccharomyces boulardii (500-1000mg/day according to age) led to the rapid elimination of symptoms in 18 of the children and negativation of the Clostridium toxin in 16 of them3.
Treatment of acute diarrhoea
In both children and adults, diarrhoea can often start violently and last anything from a couple of days to a few weeks. Severity depends on its aetiology which may be food or other poisoning, or a bacterial, viral or parasitic infection. A number of studies have been conducted to evaluate the benefits of Saccharomyces boulardii in treating acute diarrhoea.One such study involved 27 otherwise healthy children (15 boys and 12 girls), aged between 6 months and 10 years, hospitalised for acute diarrhoea. Half were given 250mg of S. boulardii dissolved in 5ml water, twice a day for 7 days, and the other half a placebo. Results confirmed the efficacy of S. boulardii against acute childhood gastroenteritis, indicating that the treatment boosts the immune response4. Another study on 100 child outpatients aged between 3 and 24 months with mild to moderate acute diarrhoea demonstrated that S. Boulardii as an adjunct treatment to oral rehydration reduced the duration of the diarrhoea, accelerated recovery and lowered the risk of prolonged diarrhoea. Efficacy was greater when S. boulardii was administered within the first 48 hours of disease5.
Travellers’ diarrhoea
The efficacy of S. boulardii in preventing travellers’ diarrhoea was investigated in a double-blind, placebo-controlled study on 1231 travellers. Subjects were divided into three groups, receiving daily either 250mg of S. boulardii, 500mg of S. boulardii, or a placebo. Treatment began five days before the start of their travel and continued for the duration of their trip. The preventive effect depended on the areas visited and was slightly greater with a higher dose. It reduced incidence of diarrhoea by between 20 and 25 %6.Treatment of chronic diarrhoea in HIV patients
In HIV patients, diarrhoea can be caused by various infectious aetiologies but in a large proportion of cases, the origin remains unknown. The resulting loss of nutrients, electrolyte imbalance and dehydration are particularly dangerous in immunosuppressed patients. In an open-label trial7, 17 seropositive patients with chronic diarrhoea were given S. boulardii for 15 days. At the end of the treatment, the number of stools decreased from 9 to 2 a day and patients regained an average 4kg in weight during the study. A double-blind, placebo-controlled, week-long study enrolled 35 HIV seropositive patients suffering from chronic diarrhoea. At the end of the seven days, the diarrhoea had gone in 56% of the patients taking S. boulardii compared with only 6% of those taking a placebo8.Improves symptoms of irritable bowel syndrome
Irritable bowel syndrome manifests as lower abdominal pain, diarrhoea and/or constipation and bloating. In a randomised, double-blind, placebo-controlled study, 34 patients - all suffering from diarrhoea - received 250mg of S. boulardii three times a day for a month. Both doctors and patients assessed the treatment as being statistically superior to placebo. The number of stools decreased and their consistency improved 9.References:
1. Adam M. et al., Controlled double blind clinical multicentre trials od Saccharomyces boulardii, Medecine & Chirurgie digestives, 1976, 5(6); 401-406.
2. Surawicz C.M. et al., Treatment of recurrent Clostridium difficile colitis with vancomycin and Saccharomyces boulardii, Am. J. Gastroenterol., 1989 Oct, 84(10): 1285-7.
3. Buts J.P. et al., Saccharomyces boulardii for Clostridium difficile-associated enterocolopathies in infents, J. Pediatr. Gastroenterol. Nutr., 1993, 16: 419-25.
4. Ozkan T.B. et al., Effect of Saccharomyces boulardii in children with acute gastroenteritis and relationship to the immune response, J. Int. Med. Res., 2007 Mar-Apr, 35(2): 201-12.
5. Villaruel G. et al., Saccharomyces boulardii in acute childhood diarrhoea: a randomized, placebo-controlled study, Acta Paediatr., 2007 Apr, 96(4): 538-41, e-pub 2007 Feb 14.
6. Kollaritsch H.H. et al., Prophylaxe der Reisediarrhoe. Ergebnisse einer doppelblinden, plazebokontrollierten Studie über die Wirsamkeit von Saccharomyces boulardii, Münch med Wschr., 38, 1988, 671-674.
7. Saint-Marc T. et al., Efficacité de Saccharomyces boulardii dans le traitement des diarrhées du sida, Annales de médecine interne, 1991, 142, 64-65.
8. Saint-Marc T. et al., Diarrhées en relation avec le sida. Essai en double aveugle de Saccharomyces boulardii, Sem. Hôp. Paris, 1995, 23-24.
9. Drossmann D.A., An integrated approach to the irritable bowel syndrome, Aliment Pharmacol. Ther., 1999, 13, suppl 2; 3-14.
Order the nutrient mentioned in this article

Standardised 2 x 1010 CFU/g. Probiotic used for decades for treating diarrhoea
www.supersmart.comFurther reading
10-10-2016
In India, turmeric is used to treat a wide variety of ailments including gastrointestinal problems, inflammation, headaches, infections and colds. It is turmeric’s curcuminoid content,...
Read more22-02-2017
Louis XIV famously suffered from severe constipation and his doctors would regularly ask him: ”Comment allez-vous (How are you)?”, when what they actually meant was:...
Read more29-01-2018
Every year, more than 750,000 people die from stomach cancer, one of the five most deadly forms of the disease. The main culprit is a...
Read more© 1997-2026 Fondation pour le Libre Choix
All rights reserved
All rights reserved
Free
Thank you for visiting our site. Before you go
REGISTER WITHClub SuperSmart
And take advantage
of exclusive benefits:
of exclusive benefits:
- Free: our weekly science-based newsletter "Nutranews"
- Special offers for club members only