29-01-2018
Helicobacter Pylori: information on the silent bacteria that kills 600,000 people a year
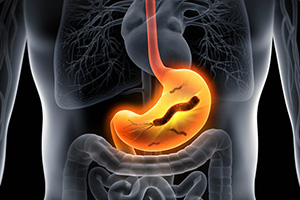 Every year, more than 750,000 people die from stomach cancer, one of the five most deadly forms of the disease. The main culprit is a pathogenic bacteria, discovered just 40 years ago, which colonises the stomach and is thought to be present in 50% of the world’s adult population. Has it penetrated the inner layers of your stomach? Will you be among the next victims?
Every year, more than 750,000 people die from stomach cancer, one of the five most deadly forms of the disease. The main culprit is a pathogenic bacteria, discovered just 40 years ago, which colonises the stomach and is thought to be present in 50% of the world’s adult population. Has it penetrated the inner layers of your stomach? Will you be among the next victims?
Before 1982, it was believed to be impossible for a living organism to grow in a such a hostile environment as the stomach. Yet two scientists studying the stomach wall in ulcer patients discovered the existence of a hitherto unknown bacteria. Before long, this microorganism was being linked to various gastric problems including stomach cancer, and its causative role was indeed proven in 1994: the International Agency for Research on Cancer categorised Helicobacter Pylori as a Group 1 carcinogen, which means it is associated with a definite risk of cancer in humans. Today, there appears to have been a staggering rise in victims: between 500,000 and 700,000 people succumb each year as a result of the slow colonisation of their stomachs. What’s even more worrying is that one in two of us may be infected!
How does Helicobacter Pylori cause cancer?
How does this bacteria manage to survive the harsh acidic environment of the stomach? And how can it remain undetected for years before suddenly triggering cancer? With the publication of the very latest research in December 2017, it seems scientists are beginning to provide precise answers to these two questions.
Helicobacter Pylori is a bacteria which lives exclusively in the human stomach. With its motile flagella, it is able to move around in gastric fluid and pass easily through the mucus that coats the stomach wall. It survives in the stomach due to the production of urease, a protein which neutralises the acid environment by converting urea into ammonia1. Once in the stomach, the bacteria secretes an enzyme (HtrA) which is able to split the bonds between gastric mucosa cells, bonds which safeguard the impermeability of the stomach’s protective lining. Any breakage thus enables the Helicobacter pylori to breach the defences and establish itself in the innermost layers of the stomach, normally inaccessible to pathogens, and begin its damaging course in complete stealth.
But how does the immune system respond?
Though the bacteria’s presence in the stomach’s inner layers sounds the alarm, triggering chronic inflammation, Helicobacter pylori has other tricks up its sleeve. In binding to the host cells of these inner layers, it injects cytotoxin-associated protein (CagA), which is able to reprogram the way the cells function in order to prevent the immune system from recognising and eliminating them2. This is a crucial step: with a blatant disregard for the body’s immune defences, it enables the bacteria’s proliferation and long-term survival. Over time, it is also responsible for converting the cells into tumour cells: CagA initiates a multitude of secondary signals, some of which are associated with cancer3-5 (including expression of beta-catenin and mesenchymal-epithelial transition). This doesn’t mean that everyone infected with the bacteria will go on to develop cancer, but it does mean they have a significantly increased risk, as do smokers, those with a high consumption of red meat and salt, or those who are regularly exposed to nitrites. The big problem is that stomach cancer is often diagnosed too late for treatment to be effective, and that the best way of curing it is still to ensure there is no silent parasitic colonisation of the gastric sub-strata.
How can you tell if you’re infected?
Contrary to what the statistics would suggest, it is not that easy for the bacteria to be passed from one individual to another. In fact, contamination occurs for the most part in childhood. It can be transmitted by saliva or by sharing cutlery, following regurgitation of vomiting by the infected person. This is why transmission occurs mostly at a young age, at school or at home: infection among adults is not that common and once treated, is actually rare6.
There are various tests for confirming whether the bacteria is in your stomach. Your doctor may either use an antibody blood test, look for traces of the bacteria in your stools or detect its presence by conducting a breath test which requires you to ingest an identifying substance and then exhale into a bag.
But it’s also possible to predict its presence. As the stomach’s protective layer suffers recurrent damage from the bacteria, and the stomach’s acidity often changes, infected individuals sometimes show signs of the pathogen’s presence. Gastric fluids can attack innermost cells inadequately protected by the mucosa, causing repeated stomach aches, ulcers and chronic inflammation which manifests in gastric discomfort or heaviness (usually a few hours after eating). Helicobacter pylori toxins also provoke the immune system leading to a proliferation of lymphocytes (with no effect on the bacteria), which may potentially result in lymphoma. Most of the time, however, the signs are too vague which is what makes this bacteria so dangerous…
How can you get rid of Helicobacter pylori quickly?
The sooner you can eliminate this parasite from your stomach, the sooner you’ll stop existing damage from developing into stomach cancer. If you’ve suffered from gastric pain and discomfort for a number of years, you’re also likely to see the difference.
Conventional treatment consists of a combination of two antibiotics plus a drug for reducing stomach acid, to be taken for around two weeks7. However, over the last 20 years or so, Helicobacter pylori strains appear to have developed increasing resistance to antibiotics. The scientific community is thus redoubling its efforts to find alternatives to such treatment which in any case, can sometimes produce side-effects.
One distinct possibility which constitutes a credible alternative or complement to current treatment is supplementing with Lactobacillus reuteri, a probiotic that inhibits Helicobacter pylori. Researchers have demonstrated its ability in vitro and in vivo to both interfere with the pathogen’s adherence, and produce antimicrobial molecules that impede its growth8-9. Specifically, this probiotic reduces levels of Helicobacter pylori by combining with it to form co-aggregates which are evacuated from the stomach via the intestinal tract. This alternative approach does not affect the gut flora and prevents the bacteria from becoming resistant to treatment. Studies have also demonstrated its propensity for strengthening the epithelial barrier (which is damaged by Helicobacter pylori) and accelerating its repair10.
Those who suspect they’ve been infected by Helicobacter pylori are thus strongly advised to take a Lactobacillus reuteri supplement Such supplementation also benefits from increased intake of antioxidants (particularly vitamins C and E) from fruits and vegetables 11 and in people with a high risk of stomach cancer (those with a number of risk factors or with a family history of the disease), it can be boosted by antibiotic treatment. In a few years’ time, we’re likely to see the emergence of other treatments aimed specifically at combatting Helicobacter pylori: the CagA injected by the bacteria and the HtrA enzyme which opens cell-to-cell junctions are the two main targets.
References
1. Frédéric Fischer, Marie Robbe-Saule, Evelyne Turlin, Francesco Mancuso, Valérie Michel, Pierre Richaud, Frédéric J. Veyrier, Hilde De Reuse, Daniel Vinella. Characterization in Helicobacter pylori of a Nickel Transporter Essential for Colonization That Was Acquired during Evolution by Gastric Helicobacter Species. PLOS Pathogens, 2016; 12 (12): e1006018 DOI: 10.1371/journal.ppat.1006018
2. Nicole Tegtmeyer, Silja Wessler, Vittorio Necchi, Manfred Rohde, Aileen Harrer, Tilman T. Rau, Carmen Isabell Asche, Manja Boehm, Holger Loessner, Ceu Figueiredo, Michael Naumann, Ralf Palmisano, Enrico Solcia, Vittorio Ricci, Steffen Backert. Helicobacter pylori Employs a Unique Basolateral Type IV Secretion Mechanism for CagA Delivery. Cell Host & Microbe, 2017; 22 (4): 552 DOI: 10.1016/j.chom.2017.09.005
3. Franco, A.T., Israel, D.A., Washington, M.K., Krishna, U., Fox, J.G., Rogers, A.B., Neish, A.S., Collier-Hyams, L., Perez-Perez, G.I., Hatakeyama, M., et al. (2005). Activation of beta-catenin by carcinogenic Helicobacter pylori. Proc. Natl. Acad. Sci. USA 102, 10646–10651.
4. Murata-Kamiya, N., Kurashima, Y., Teishikata, Y., Saito, Y., Higashi, H., Aburatani, H., Akiyama, T., Peek, R.M., Jr., Azuma, T., et al. (2007). Helicobacter pylori CagA interacts with E-cadherin and deregulates the beta-catenin signal that promotes intestinal transdifferentiation in gastric epithelial cells. Oncogene 26, 4617–4626.
5. Lee, D.G., Kim, H.S., Lee, Y.S., Kim, S., Cha, S.Y., Ota, I., Kim, N.H., Cha, Y.H., Yang, D.H., Lee, Y., et al. (2014). Helicobacter pylori CagA promotes Snailmediated epithelial-mesenchymal transition by reducing GSK-3 activity. Nat. Commun. 5, 4423.
6. Brown LM. Helicobacter pylori : epidemiology and routes of transmission. Epidemiologic reviews 2000;22(2):283-97
7. Courillon-Mallet A. Résistance de Helicobacter pylori : qui traiter et comment ? Presse Med 2006 ;35 :657-62.
8. Holz C, Busjahn A, Mehling H, Arya S, Boettner M, Habibi H, Lang C. Significant Reduction in Helicobacter pylori Load in Humans with Non-viable Lactobacillus reuteri DSM17648: A Pilot Study. Probiotics Antimicrob Proteins. 2015 Jun;7(2):91-100. doi: 10.1007/s12602-014-9181-3.
9. Mehling H, Busjahn A. Non-viable Lactobacillus reuteri DSMZ 17648 (Pylopass™) as a new approach to Helicobacter pylori control in humans. Nutrients. 2013 Aug 2;5(8):3062-73. doi: 10.3390/nu5083062.
10. Lu L, Walker WA. Pathologic and physiologic interactions of bacteria with the gastrointestinal epithelium. Am J Clin Nutr 2001;73;1124S-1130S.
11. Malfertheiner P et al. Current concepts in the management of Helicobacter pylori infection : the Maastricht III consensus report. Gut 2007;56:772-81
Order the nutrients mentioned in this article

A new-generation probiotic that supports cardiovascular health
www.supersmart.comFurther reading
10-10-2016
In India, turmeric is used to treat a wide variety of ailments including gastrointestinal problems, inflammation, headaches, infections and colds. It is turmeric’s curcuminoid content,...
Read more22-02-2017
Louis XIV famously suffered from severe constipation and his doctors would regularly ask him: ”Comment allez-vous (How are you)?”, when what they actually meant was:...
Read more31-07-2017
Food cravings affect those trying to lose weight just as they do stable-weight individuals. Finding a lasting solution to these sudden ‘snack attacks’ requires a...
Read more© 1997-2026 Fondation pour le Libre Choix
All rights reserved
All rights reserved
Free
Thank you for visiting our site. Before you go
REGISTER WITHClub SuperSmart
And take advantage
of exclusive benefits:
of exclusive benefits:
- Free: our weekly science-based newsletter "Nutranews"
- Special offers for club members only